 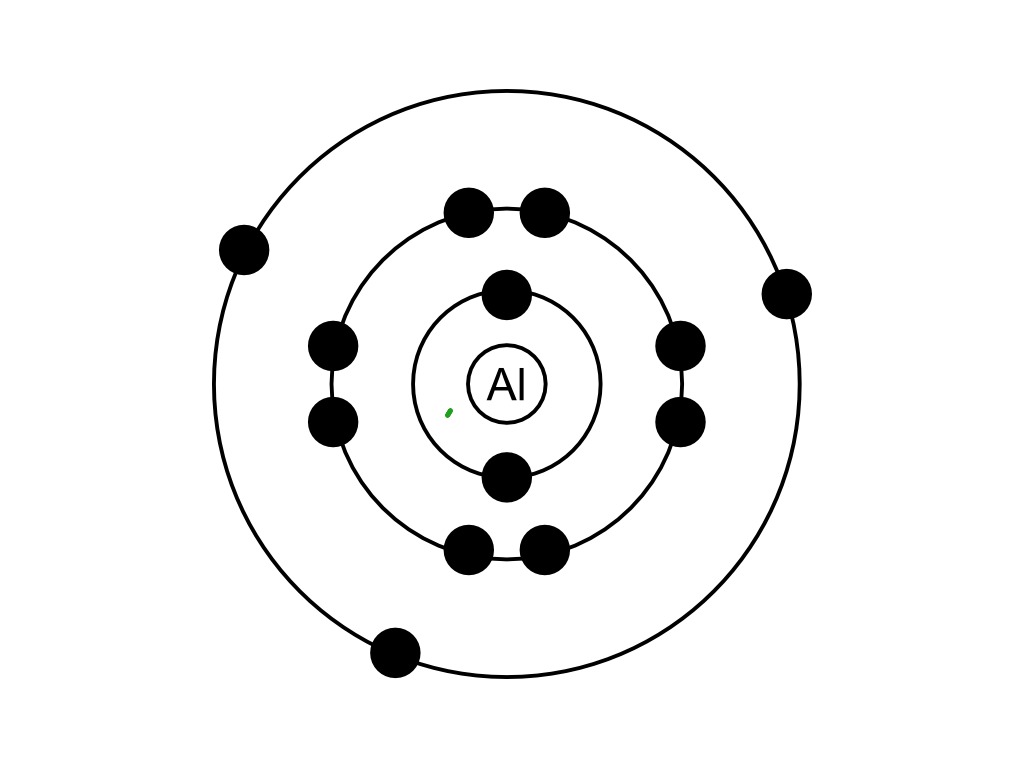
If the phosphorus is going to form PCl 5 it has first to generate 5 unpaired electrons. In this case, a more modern view makes things look better by abandoning any pretense of worrying about noble gas structures. This diagram also shows only the outer electrons. You will remember that the dots-and-crosses picture of PCl 5 looks awkward because the phosphorus doesn't end up with a noble gas structure. What's wrong with the simple view of PCl 5? You might wonder whether all this is worth the bother! Probably not! It is worth it with PCl 5, though. The four 3-level orbitals hybridise to produce 4 equivalent sp 3 hybrids just like in carbon - except that one of these hybrid orbitals contains a lone pair of electrons.Įach of the 3 chlorines then forms a covalent bond by merging the atomic orbital containing its unpaired electron with one of the phosphorus's unpaired electrons to make 3 molecular orbitals. There are 3 unpaired electrons that can be used to form bonds with 3 chlorine atoms. If we look only at the outer electrons as "electrons-in-boxes": Phosphorus has the electronic structure 1s 22s 22p 63s 23p x 13p y 13p z 1. Nothing is wrong with this! (Although it doesn't account for the shape of the molecule properly.) If you were going to take a more modern look at it, the argument would go like this: 
What's wrong with the simple view of PCl 3? This diagram only shows the outer (bonding) electrons. The bonding in the phosphorus chlorides, PCl 3 and PCl 5 It starts with a simple picture of the single covalent bond. This page explains what covalent bonding is. What's wrong with the simple view of PCl5?.The bonding in the phosphorus chlorides, PCl3 and PCl5. 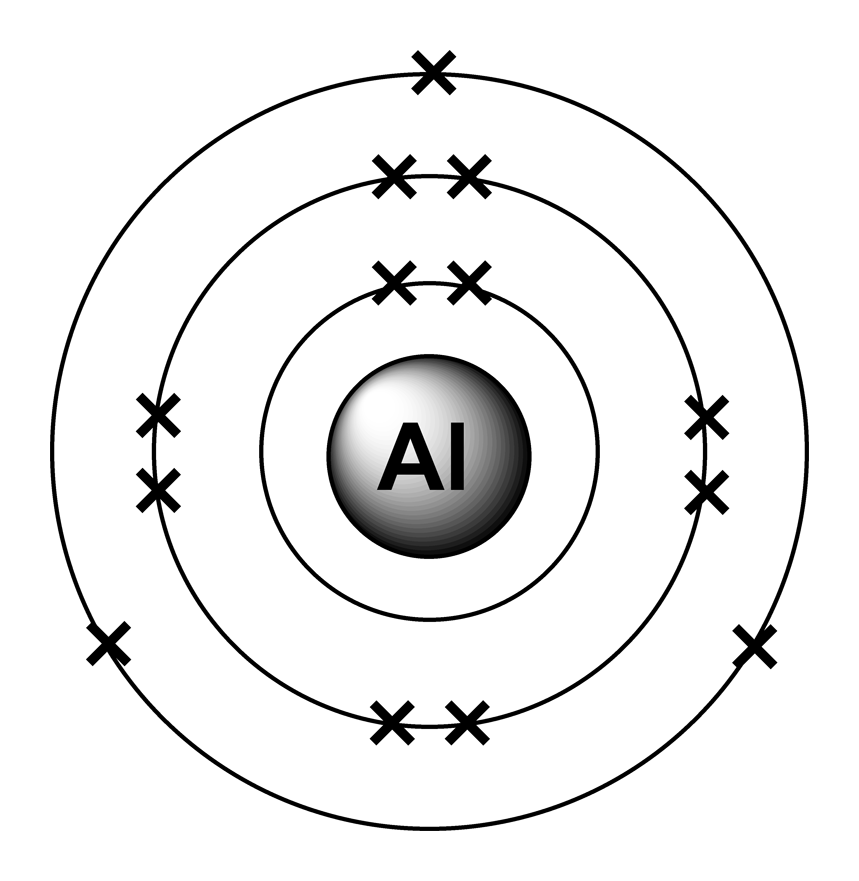
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
